Chlorine is by far the most abundant of the halogens, and the only one needed in relatively large amounts (as chloride ions) by humans. Many synthetic organic compounds such as plastic polymers, and a few natural ones, contain halogen atoms these are known as halogenated compounds or organic halides. 
Some properties, however, are found in neither parent halogen − Cl 2 and I 2 are soluble in CCl 4 but ICl is not, since it is a polar molecule due to the relatively large electronegativity difference between I and Cl. The properties and behaviour of a diatomic interhalogen compound tend to be intermediate between those of its parent halogens. BrF, ICl, ClF) bear resemblance to the pure halogens in some respects. 
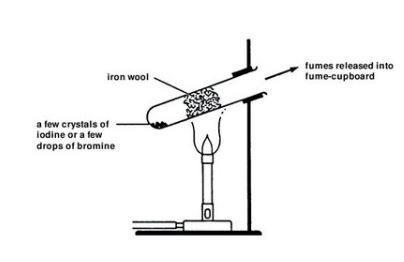
The halogens react with each other to form interhalogen compounds. HAt, or "hydrastatic acid", should also qualify, but it is not typically included in discussions of hydrohalic acid due to astatine's extreme instability toward alpha decay. When in aqueous solution, the hydrogen halides are known as hydrohalic acids. The halogens all form binary compounds with hydrogen, the hydrogen halides, HX ( HF, HCl, HBr, HI), a series of particularly strong acids. Sodium hypochlorite, which is produced from chlorine, is the active ingredient of most fabric bleaches and chlorine-derived bleaches are used in the production of some paper products. 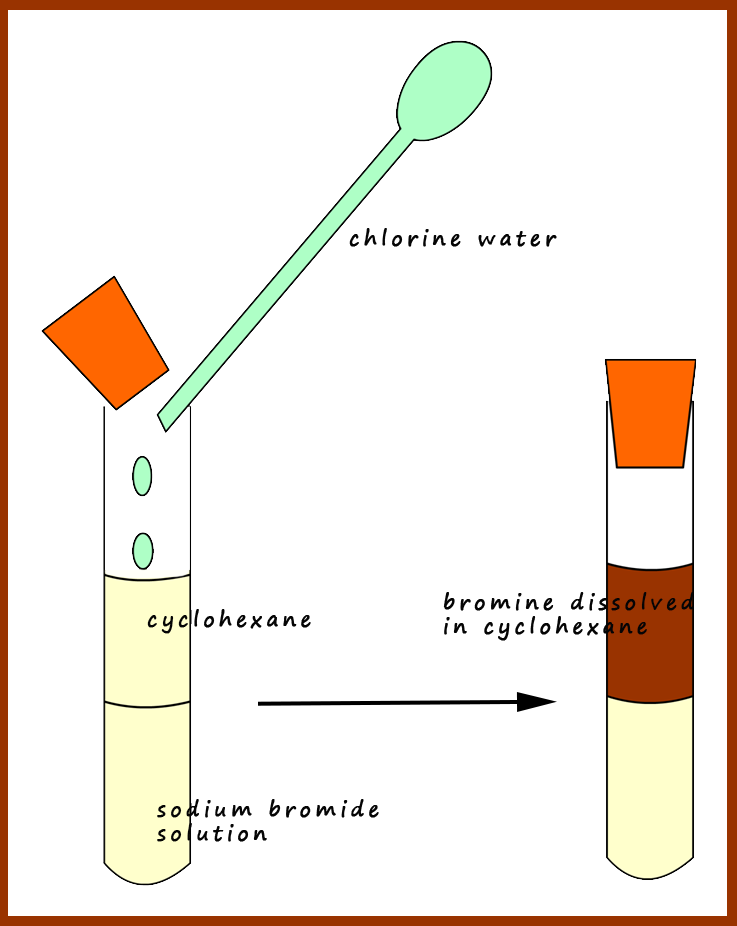
Their reactivity is also put to use in bleaching. They kill bacteria and other potentially harmful microorganisms through a process known as sterilization. Thus fluorine must be handled with substances such as Teflon, extremely dry glass, or metals such as copper or steel which form a protective layer of fluoride on their surface.īoth chlorine and bromine are used as disinfectants for drinking water, swimming pools, fresh wounds, dishes, and surfaces. The reactivity of fluorine is such that, if used or stored in laboratory glassware, it can react with glass in the presence of small amounts of water to form SiF 4. Fluorine is the most reactive element in existence, attacking such inert materials as glass, and forming compounds with the heavier noble gases. They can gain this electron by reacting with atoms of other elements. This high reactivity is due to their atoms being one electron short of a full outer shell of eight electrons. Halogens are highly reactive, and as such can be harmful or lethal to biological organisms in sufficient quantities. * Ununseptium has not yet been discovered values are either unknown if no value appears, or are estimates based on other similar chemicals. The halogens show a number of trends when moving down the group - for instance, decreasing electronegativity and reactivity, increasing melting and boiling point. The term halogen originates from 18th century scientific French nomenclature based on adaptations of Greek roots: hals (sea) or halas (salt), and gen- (to generate) - referring to elements which produce a salt in union with a metal. At room temperature and pressure, fluorine and chlorine are gases, bromine is a liquid and iodine and astatine are solids Group 17 is therefore the only periodic table group exhibiting all three states of matter at room temperature. In their elemental forms, the halogens exist as diatomic molecules, but these only have a fleeting existence in nature and are much more common in the laboratory and in industry. Halogenated organic compounds can also be found as natural products in living organisms. Halide ions and oxoanions such as IO 3 − can be found in many minerals and in seawater. Owing to their high reactivity, the halogens are found in the environment only in compounds or as ions.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
